What is Multikine®
Multikine, CEL-SCI's lead investigational immunotherapy, is an immunotherapeutic agent that is being developed as a potential first-line neoadjuvant therapy in patients with advanced primary squamous cell carcinoma of the head and neck. If it were to be approved for use following completion of CEL-SCI's clinical development program, Multikine would be a different kind of therapy in the fight against cancer; one that appears to have the potential to work with the body's natural immune system in the fight against tumors. Multikine may also have the potential to be a first in a new class of cancer immunotherapy drugs. It is the first investigational combination immunotherapy thought to have both passive and active immune properties and has displayed the following characteristics in studies to date:
- Potential first-line treatment: Multikine is administered prior to any other therapy because CEL-SCI believes that this is the period when there is a greater potential likelihood of activating an anti-tumor immune response. Once the patient has had surgery or has received radiation and/or chemotherapy, the immune system may become weakened and may be less able to mount an anti-tumor immune response.
- Potential to work with the body: Multikine investigational immunotherapy is directed at activating and boosting the body's own anti-tumor immune response to potentially help the body in addressing the tumor.
- A mixture of different immune molecules, potentially simulating the natural immune system: Multikine is composed of a defined mixture of naturally occurring molecules which in early-phase studies have been reported to be biologically/immunologically active.
Multikine
Multikine (Leukocyte Interleukin, Injection) is the full name of this investigational therapy, which, for simplicity, is referred to in the remainder of this page as Multikine. Multikine has not been licensed or approved by the FDA or by any other regulatory agency. Similarly, its safety or efficacy has not been established for any use.
In the Phase III clinical trial, Multikine is administered to cancer patients prior to their receiving any treatment for cancer, including surgery, radiation and/or chemotherapy
Clinical Trial Status
The first indication we are pursuing for our Multikine investigational immunotherapy product candidate is for the use as a neoadjuvant therapy in patients having advanced primary (not yet treated) squamous cell carcinoma of the head and neck, SCCHN. SCCHN represents about 90% of head and neck cancer, and CEL-SCI believes that there is a large, unmet medical need among head and neck cancer patients. To the best of our knowledge, the last FDA approval of a therapy indicated for the treatment of advanced primary head and neck cancer was decades ago. Head and neck cancer represents approximately 890,000 patients diagnosed worldwide each year, and nearly 60,000 patients diagnosed annually in the United States. Multikine investigational immunotherapy was granted Orphan Drug designation for neoadjuvant therapy in patients with SCCHN by the FDA in the United States.
Read More

Phase III Trial Design
The Phase III Clinical Trial: Multikine in SCCHN patients was cleared and received a go-ahead from 24 Regulatory Agencies worldwide including the U.S., Canada, France and the United Kingdom. The Phase 3 study accrued its first patient in early 2011 and achieved complete enrollment in September 2016. The study ended in the spring of 2020, the database was locked in December 2020 and topline results were released in June 2021. The Phase III study in newly diagnosed advanced primary head and neck cancer with our Multikine immunotherapy produced a statistically significant 5-year survival benefit in patients receiving surgery plus radiotherapy, representing approximately 40% of the study population, and it is estimated that globally about 210,000 patients annually could potentially benefit from it should it be approved. The 5-year overall survival (OS) benefit in this study treatment arm was robust and durable, with no safety issues, something not commonly seen with cancer drugs. In fact, the survival benefit increased over time and at 5-years the overall survival benefit reached an absolute 14.1% advantage for the Multikine treated arm over control (n=380, total study patients treated with surgery plus radiation), Multikine arm 62.7% and control arm 48.6%, survival.
- Total Number of Subjects Enrolled in the Phase III: 928
- Clinical Centers: approximately 100 globally distributed; North America, Europe, Asia
- Total Number of Subjects Enrolled in the Phase III: 928
- Clinical Centers: approximately 100 globally distributed; North America, Europe, Asia
For more information about the Phase III Study please see www.clinicaltrials.gov (NCT number: NCT01265849)
Read MoreStudy Title/Summary: A Pivotal Phase III, Open-label, Randomized, Controlled Multi-center Global Study of the Effects of Multikine Plus Standard of Care (Surgery + Radiotherapy or Surgery + Concurrent Chemoradiotherapy) in Subjects with Advanced Primary Squamous Cell Carcinoma of the Oral Cavity and Soft Palate Versus Standard of Care Only.
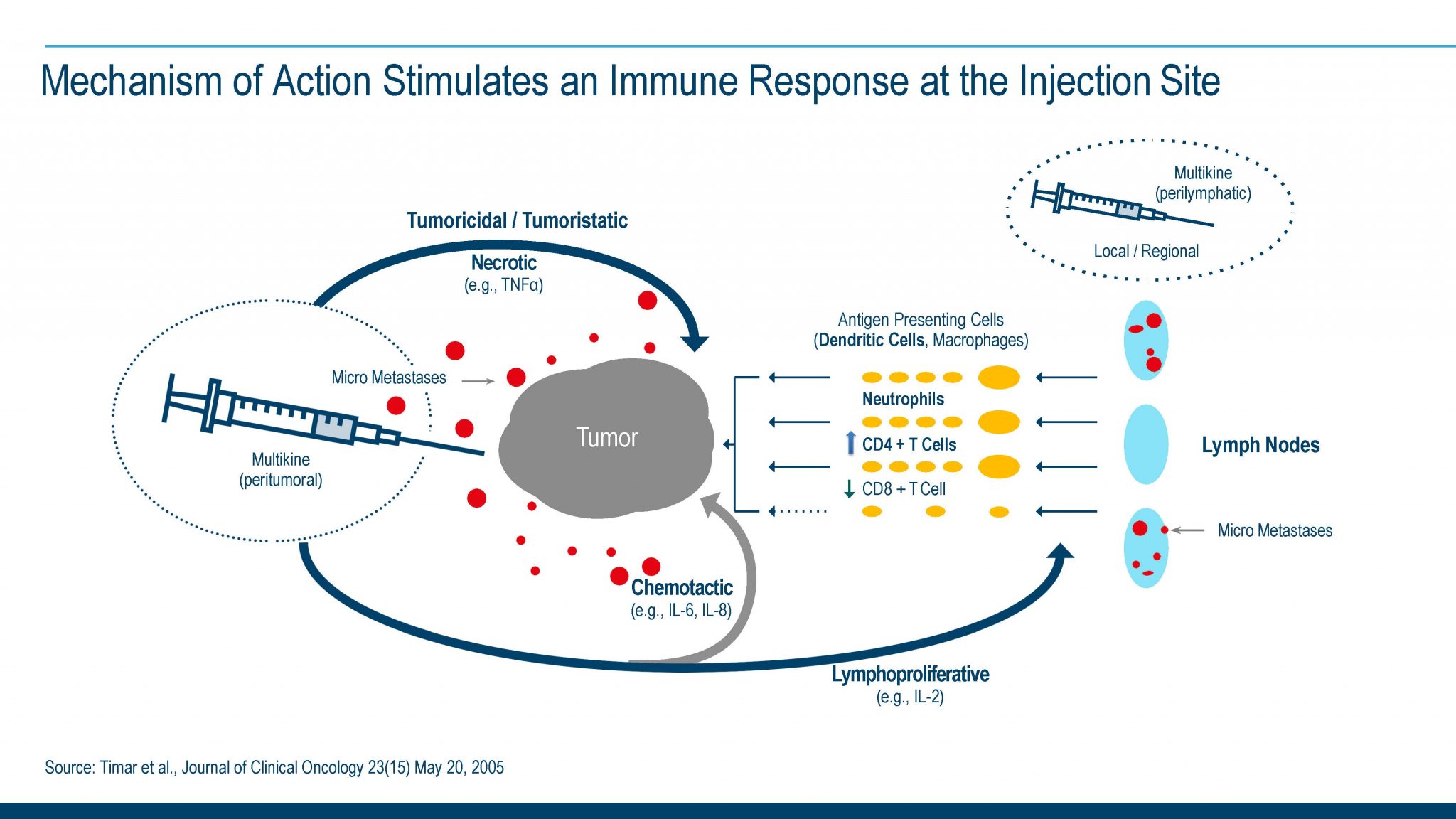
Current Understanding of the Potential Mechanism of Action of the Investigational Therapy Multikine (Leukocyte Interleukin, Injection)
Mechanism of Action
Clinical and pathology data from Phase I and Phase II clinical trials with Multikine investigational immunotherapy published in peer reviewed scientific journals (Feinmesser et al Arch Otolaryngol. Surg. 2003; Timar et al 2003 and 2005) suggest that Multikine has the potential to produce an anti-tumor response, as the reported data from these early-stage studies suggests:
- Multikine potentially may recognize and/or bind to multiple (different) antigens (or receptors) on the cancer cells.
- Multikine potentially may directly affect/kill cancer cells: The various cytokines present in the Multikine investigational therapy, such as TNF, IL-2, IFN, along with other cytokines, could be responsible for this potential activity.
- Multikine may signal the immune system to produce an anti-tumor immune response: Clinical data (reported from Phase II studies) also suggest that Multikine could augment the type of cells that infiltrate and attack the tumor changing the tumor microenvironment ratio of CD4/DC8 cells from CD-8 cells to predominantly CD-4 cells. These CD-4 cells have the potential to bring about an anti-tumor immune response (Timar et al 2005).
Multikine may have the potential to render the remaining cancer cells susceptible to radiation treatment (Timar et al 2003).
Read More
Potential indications/
relevance to the medical community
The first indication we are pursuing for our Multikine product candidate is for the use as first-line neoadjuvant treatment in newly diagnosed patients with advanced primary squamous cell carcinoma of the head and neck, SCCHN. SCCHN is a type of head and neck cancer. In Phase I and Phase II clinical trials and in the recently completed Phase III clinical trial, Multikine was administered right after diagnosis and prior to any other cancer therapy because CEL-SCI believes that this is the period when there is a greater likelihood of activating an anti-tumor immune response. The Phase III study in newly diagnosed advanced primary head and neck cancer with our Multikine immunotherapy produced a statistically significant 5-year survival benefit in patients receiving surgery plus radiotherapy, representing approximately 40% of the study population and it is estimated that globally about 210,000 patients annually could potentially benefit from it should it be approved. The 5-year overall survival (OS) benefit in this study treatment arm was robust and durable, with no safety issues, something not commonly seen with cancer drugs. In fact, the survival benefit increased over time and at 5-years the overall survival benefit reached an absolute 14.1% advantage for the Multikine treated arm over control (n=380, total study patients treated with surgery plus radiation), Multikine arm 62.7% and control arm 48.6%, survival.
Read More

