Introduction and overview
L.E.A.P.S.(TM)* is a patented, T-cell modulation, peptide epitope delivery technology that enables CEL-SCI to design and synthesize proprietary peptide immunogens. L.E.A.P.S.(TM) compounds consist of a small T-cell binding peptide ligand linked with a disease-associated peptide antigen.
This platform technology has been shown in several animal models to preferentially direct immune response to a cellular (e.g. T-cell), humoral (antibody) or mixed pathway. It has the potential to be utilized in diseases for which antigenic epitope sequences have already been identified, such as: a number of infectious diseases, some cancers, autoimmune diseases, allergic asthma and allergy, and select CNS diseases (e.g., Alzheimer’s).
L.E.A.P.S.(TM) Overview
L.E.A.P.S.(TM) is a T-cell modulation platform technology that enables CEL-SCI to design and synthesize proprietary immunogens. Diseases for which an antigenic epitope sequence has been identified such as: a number of infectious diseases, some cancers, autoimmune diseases, allergic asthma and allergy, and select CNS diseases all represent potential therapeutic or preventive candidates for development using the application of L.E.A.P.S.(TM) technology.
The L.E.A.P.S.(TM) compounds are thought to be bi- or hetero- functional peptides, including an antigenic peptide (Ag) linked to another peptide referred to as a T-cell binding ligand (TCBL). This complex may allow the presentation of antigen to occur at the same time as delivery of a co-stimulatory signal. Several types of TCBLs have been evaluated. Two of particular interest are used and are designated TCBL peptides J and G or the changed version of G, CEL-1000. Conjugates of these appear to activate different sub-sets of T-cells. J is a short fragment from b-microglobulin (aa38-50) and G and CEL-1000 are a modified fragment from MHC II b-chain (aa135-149). Based on site directed mutagenesis studies of MHC II b-chain and/or peptide competition studies, G presumably binds to CD4, a T cell co-stimulator molecule.
The concept behind the L.E.A.P.S.(TM) technology is to directly mimic cell/cell interactions on the T-cell surface with synthetic peptides. The L.E.A.P.S(TM) constructs containing the antigenic disease epitope linked to a T-cell binding ligand (TCBL) can be manufactured by peptide synthesis or by covalently linking the two separately synthesized peptides. In animal studies CEL-SCI has demonstrated that depending on the type of L.E.A.P.S.(TM) construct and TCBL used, it is possible to create peptides that when administered to animals (in the setting of animal studies) these peptide constructs are able to direct the immune response outcome towards the development of T-cell function with primarily effector T-cell functions (T Lymphocyte; helper/effector T lymphocyte, type 1 or 2 [Th1 or Th2], cytotoxic [Tc] or suppressor [Ts]). Therefore, it would appear that L.E.A.P.S.(TM) construct represents a chimeric peptide with bi-functional behavior (as depicted in the Figure below).
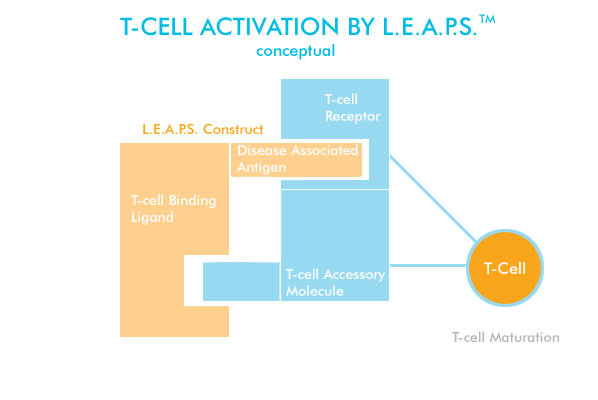
Schematic representation of a L.E.A.P.S.(TM)
construct and a receptor on the T cell
No L.E.A.P.S. product has been approved for sale, barter or exchange by the FDA or any other regulatory agency for any use to treat disease in animals or humans. The safety or efficacy of these products has not been established for any use. Lastly, no definitive conclusions can be drawn from these early-phase, preclinical-trials data involving these investigational products.
* L.E.A.P.S. is the trademark of CEL-SCI for this investigational platform technology, and this proprietary name is subject to FDA review.

